研究内容
私たちの研究活動の一端を動画や写真、イラストを交えながら、ご説明したと思います。
ポリマーブラシとは? About "Polymer Brush"
ヒモ状の高分子を材料表面に密に生やし、歯ブラシのような構造にした分子組織を「ポリマーブラシ」と呼びます。表面から生えたポリマーブラシは、厚み10万分の1ミリ(100 nm前後)の膜として材料表面を覆います。
例えば、イオン性基を結合したポリマーブラシの表面は、超親水性を示し、濡れ性が向上します。すると、水中での油汚れ防止(防汚性)、生体適合性、低摩擦性(潤滑性)など優れた機 能が表面に発現します。また、ブラシ表面同士を貼り合わせると接着させることができ、条件を変えると剥がすこともできます。このように、ポリマーブラシは様々な分野への応用が期待されています。
An assembly of polymer chains grafted on a surface is called a “polymer brush” because those form a brush-like thin layer with thickness of approximately 100 nm.
Surfaces of polymer brushes containing ionic functional groups are show super-hydrophilicity due to improving of the surface wettability. Therefore, the surfaces exhibit remarkable properties in water, such as oil-stain repelling (antifouling), protein-adsorption resistance (biocompatibility), and low coefficient of kinetic friction (lubricity).
Moreover, two surfaces grafting polymer brushes can be adhered by attaching with facing each other, and also can be detached by changing ambient conditions.
The polymer brushes are expected to be applied in a wide range of fields.
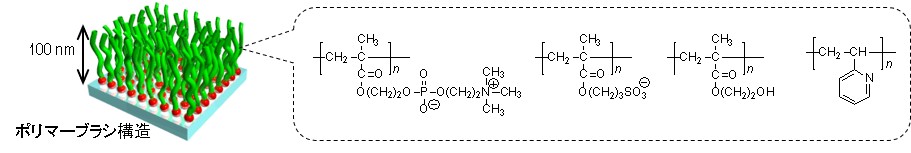
ポリマーブラシのイメージイラストと化学構造式
研究室ギャラリー
実験風景や観察された画像、研究に関わるイメージイラストを集めました。
銅触媒の還元反応 ―ポリマーブラシの調製のために―
Reduction of Cu catalyst for preparation of polymer brush by AGET ATRP
We are going to introduce a polymerization method that is commonly used in our laboratory to prepare polymer brushes.
初めに重合溶液10 mLと重合開始剤固定化シリコン基板を入れ、密閉した試験管を用意しました。この重合溶液は、メタノールと水の混合溶媒に、触媒である臭化銅(II) CrBr2、その配位子の2,2'-ビピリジル、そしてモノマーとしてスルホン酸基を有するメタクリル酸誘導体を溶解させることで調製しました。
ここに注射針が差し込まれており、気泡が発生しているのが見えると思います。これは溶液中に窒素を供給し、溶存酸素を除去するために行っています。あらかじめ、この窒素バブリングを30分間行いました。
そして、この動画で見えるように、別の注射針を上から差し込みアスコルビン酸水溶液(無色透明)を加えました。すると溶液の色は薄い青色から、赤褐色へと速やかに変わり、劇的な化学反応が起きました。これは次の半反応式のように、アスコルビン酸により銅が2価から1価に還元されたからです。
Cu2+ + e− → Cu+
このように系内で生成した1価の銅が、先ずシリコン基板に結合している開始剤を活性化します。次に、この開始剤が次々とモノマーと反応します。こうして基板からポリマーブラシが生長します。
実は、アスコルビン酸は「ビタミンC」とも呼ばれているとても身近な還元性の物質です。このような還元剤を用いることで、不安定な1価の銅を扱う必要がなくなり、重合操作が非常に簡便になります。
この重合方法は、「電子移動により生成させた活性化剤 (Activators Generated by Electron Transfer : AGET)を用いた原子移動ラジカル重合 (Atom Transfer Radical Polymerization : ATRP)」と呼ばれています。
当研究室では、こちらでご覧いただいたAGET ATRPを用いて、様々な親水性ポリマーブラシを調製することができます。そのブラシの特性について、個体表面の濡れ性をはじめ、接着、凝着力そして水中潤滑性など幅広い分野で研究を行っています。
A reaction mixture 10 mL for the polymerization and a silicon substrate immobilized a polymerization initiator was added in this sealed test tube. This polymerization solution was prepared by dissolving copper(II) bromide CrBrCrBr2 as a catalyst, its ligand 2,2'-bipyridyl, and a methacrylic acid-type monomer containing a sulfonic acid group in a mixed solvent of methanol and water.
You can see an injection needle which was inserted in the solution and generated bubbles from its head. In this way, bubbles of nitrogen were provided to remove dissolved oxygen in the solution. This nitrogen bubbling had been preliminarily performed for 30 minutes.
Then, as you can see in this video, we inserted another injection needle from above and added an ascorbic acid aqueous solution (colorless and transparent). Immediately, the solution color changed from light blue to reddish brown. This is because the ascorbic acid reduced copper from divalent Cu2+ to monovalent Cu+ as written in a following half reaction.
Cu2+ + e− → Cu+
Firstly, the in-situ generated Cu+ activates the initiator immobilized on the silicon substrate. Then, this initiator reacts with the monomers one after another. This chain propagation produces the polymer brush grafting from the substrate.
In fact, the ascorbic acid is a very familiar reducing substance, also known as “vitamin C”. Using this reducing agent extremely simplify an experimental procedure because of avoiding the trouble of handling the unstable monovalent Cu+.
This polymerization method is called “Activators Generated by Electron Transfer (AGET)” for “Atom Transfer Radical Polymerization (ATRP)”.
Our laboratory can prepare various hydrophilic polymer brushes using AGET ATRP. We conduct a study on the unique characteristics of the polymer brush in a wide range such as the wettability, adhesion, adhesion, and lubrication.
生きたフジツボ幼生の付着力測定
キプリス幼生に対する試料表面の付着防止性能をより短時間で定量的に評価する方法として、走査プローブ顕微鏡(SPM)の活用を検討してきました。こちらの動画のように、生きたアカフジツボキプリス幼生をカンチレバーに固定することにより、キプリス幼生の触角とポリマーブラシ表面や単分子膜表面との凝着力を海水中で定量することが可能となりました。
ポリマーブラシの濡れ
空気中で親水性のポリマーブラシ表面に水滴を付着させると、水は素早く濡れ広がり非常に低い接触角を示します。このようにポリマーブラシには超親水性を示す種類があります。しかし、その濡れの詳細な理論はよくわかっていません。私たちの研究室では、先行薄膜や水和水に注目してポリマーブラシの濡れを理解しようと研究を行っています。(動画)
汚れにくい表面 (水中防汚性)
Stain-resistant surface — Antifouling in Water
表面にひも状の高分子を密集して生やすとポリマーブラシという薄膜ができます。特に親水性ブラシの表面は、水中で油汚れをはじく機能や、微生物が付着しないという特徴があり、防汚性に役立ちます。
Grafting technics of chain-like polymers on a surface provide the thin layer which is called the polymer brush. In particular, the hydrophilic polymer brush surfaces exhibit remarkable antifouling properties in water: such as repelling of oil stains and preventing of micro-organism settlements.
親水性のPoly(SPMK)ブラシを陶器表面にグラフトし、この表面にラー油を1滴付着させました。これを水中に沈めると、このハイスピードカメラの映像のように、ラー油は球状になり浮かび上がりました。このように、ポリマーブラシ表面は優れた防汚性を示します。

新規ポリマーの合成

環境に優しい接着
Eco-friendly Adhesion
ポリマーブラシ同士を少量の水とともに貼り合わせると接着させることができます。それだけでなく、接着界面を再び剥がすこともできます。このような新しい接着法は、有機溶剤を使わないので環境に優しく、医療にも応用が期待されています。
Two surfaces grafting polymer brushes can be adhered by attaching each other with a small amount of water, and also can be detached by changing ambient conditions. The new adhesion technique is eco-friendly because of organic-solvent free and be expected to be utilized for medical treatments.
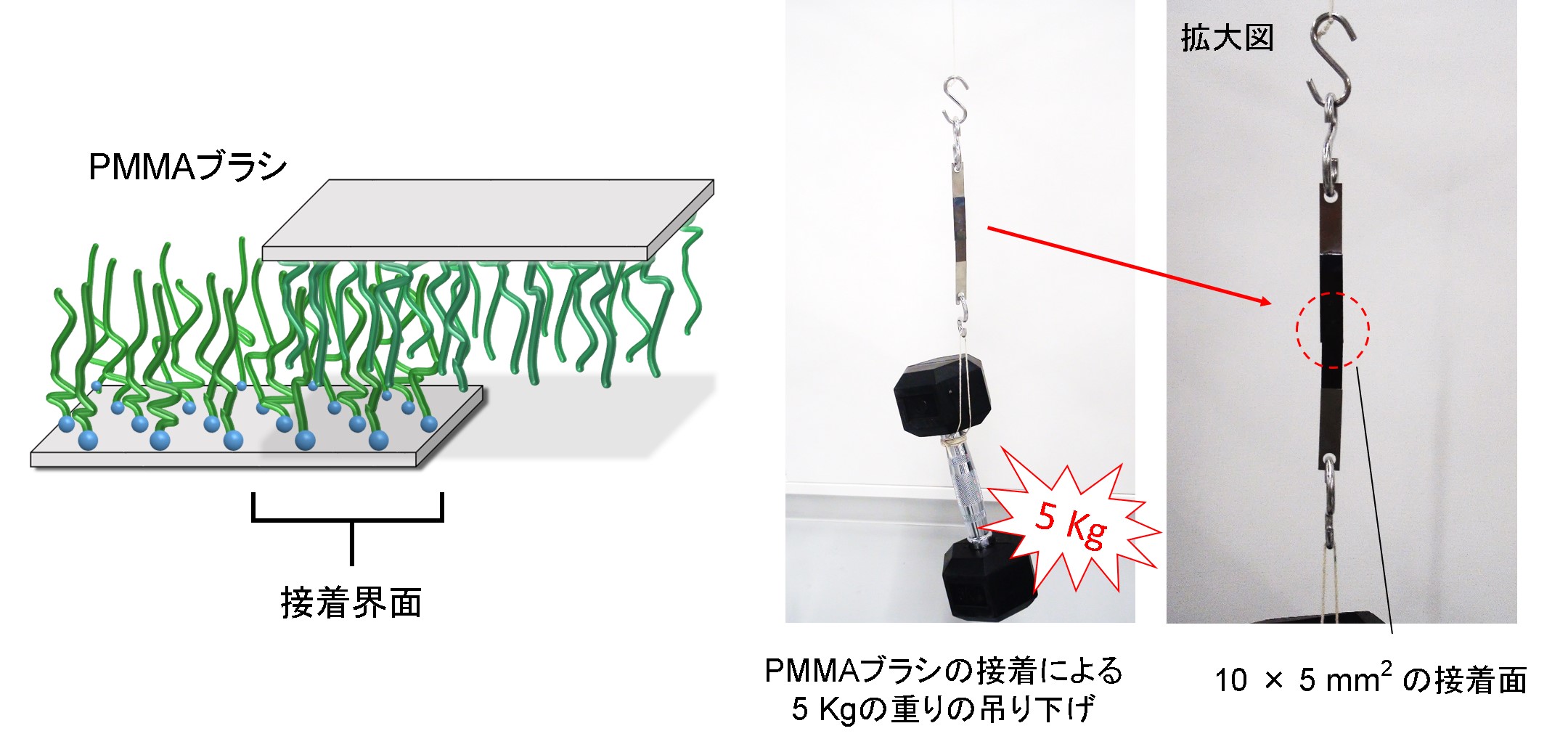
ポリマーブラシを用いた接着。10 × 5 mm2 の面積で5 kg以上の荷重に耐えることができます。

ポリマーブラシによって接着された基板同士は、どのくらいの面積で密着しているのでしょうか。観察された色と縞模様から計測を試みました。
よく滑る表面 (摩擦と潤滑)
Slippery surface — Friction and Lubrication
親水性のポリマーブラシは優れた潤滑性を示します。生体親和性の良い分子と組み合わせることで人工関節やカテーテルへの応用が期待されています。
Hydrophilic polymer brushes show excellent lubrication properties. The brushes consisting of biocompatible molecules are expected to be applied to coating on surfaces of catheters and artificial joints.
アピールポイント Strengths
様々な材料表面にポリマーブラシを調製し、表面改質を施すことができます。
ポリマーブラシを調製可能な表面の例:ガラスやシリコンの他に、ステンレス、アルミナ、チタンなどの金属、ポリエチレン、ポリカーボネート、ポリ乳酸、ナイロン、ABS樹脂、ポリイミドなどの高分子材料、木材や繊維表面。また、シリカ微粒子などの粒子にポリマーブラシを付与することで分散性や相溶性の向上を図ることができます。
For surface modification, the polymer brushes can be prepared on various surfaces:
glass, silicon, alumina, metals – stainless and titanium, polymer materials – polyethylene, polycarbonate, polylactic acid, nylon, ABS resin, and polyimide, wood materials, and fibrous materials.
In addition, particles such as silica nanoparticles can be functionalized by grafting the polymer brushes for the purpose of improving the dispersibility and the compatibility to solvents.
利用・用途 / 応用分野 Application and purpose
- 超親水性表面 Super-hydrophilic surface
- 水中防汚性 Antifouling in water
- 海洋付着生物忌避 Repellent against sessile marine organisms
- 微粒子分散 Dispersion of nanoparticles
- ブラシによる接着 Adhesion of polymer brushes
- 気泡のつかない表面 Anti-capturing air-bubbles
- 生物模倣表面 Biomimetics surface
- 電化製品の防汚処理 Antifouling treatment of electronic devices
- 太陽電池表面の防汚処理 Antifouling treatment of solar cells
- フジツボ付着抑制 船底塗料 Antifouling ship-bottom paint against barnacle settlements
- カテーテル表面のコーティング Coating on catheter surfaces
- 水潤滑と人工関節 Water lubrication and artificial joints
関連情報 Related information
-
知的財産権 | 特願2011-545211,他.
Intellectual property | Japan patent JP2011-545211, etc. -
関連論文
Journal Article
M. Kobayashi et al., Langmuir 34, 10276-10286 (2018).
Polym. J. 50, 101-108 (2018).
Polymer 119, 167-175 (2017). RSC Adv. 6, 86373-86384 (2016).
Polymer 89, 128-134 (2016).
Langmuir 28, 7212-7222 (2012).
J. Biom. Sci. Polym. Ed. 25, 1673-1687 (2014).
ACS Appl. Mater. Interfaces 6, 20365-20371 (2014).
最近の研究成果は、 査読付論文、 研究発表 (工学院大学HP 小林元康 教授プロフィールへ)からご覧になれます。 -
メディア関連 | NHK総合「凄ワザ!:汚れないまな板」2017年1月14日20:15-20:45放送
Media Coverage | NHK General TV, “Sugo-waza! The Antifouling Cutting Board” broadcasted on January 14, 2017, at 8:15-8:45 p.m.
研究テーマ
ポリマーブラシによる接着
- ポリメタクリル酸メチルブラシの熱接着における一次構造依存性
- ポリ(4-ビニルピリジン)ブラシとの水素結合を利用した接着界面の調製
- ラマン分光によるポリマーブラシ界面における分子鎖構造解析
- 低Tgポリマーブラシによる異種材料接着
表面改質・解析
- 表面開始アニオン重合による表面グラフトエラストマーの合成
- 表面グラフト化イオン性ポリマーによる水中摩擦特性制御
- 金属表面における親油性ポリマーブラシ薄膜の調製と摩擦特性
- ドーパミン型表面開始剤を用いた高分子材料の表面親水化法の検討
- 走査フォース顕微鏡による生体キプリス幼生付着器官のポリマーブラシ表面における凝着力評価
- 微細パターン化イオン性ブラシ表面における水の薄膜の可視化
双性イオン型電解質ポリマーブラシ
- インバースホスホリルコリン型双性イオンポリマーの合成と分子間相互作用
- メタクリル酸誘導体スルホベタイン型ポリマーブラシの修飾シリカ微粒子による凝着力測定
高分子の精密制御合成
- 電子供与性基を有するスチレン誘導体の制御ラジカル重合におけるプロトン性溶媒の添加効果